Once upon a time, there was a village called Hambry, which was the capital of the barony of Mejis.
Hambry was located along the coast of the Clean Sea. The village grew rich and prosperous from the fish and crude oil it extracted from the Clean Sea. The fishing industry and oil refineries along the coast were successful year-round. Business was good… for a while.

While the fishing industry factories implemented stringent rules to prevent overfishing, oil refineries performed stingy and unreliable procedures to ensure environmental safety in order to cut budgets. The sulfur removal sector specializing in the separation of sulfuric acid from petroleum were most responsible for the events that unfolded.

Air pollutant and released wastewater levels skyrocketed, but perhaps most detrimental of all was the sulfuric acid byproducts released in massive amounts to the Clean Sea (ironic, isn’t it?). These acidic byproducts affected the coastal wildlife, fishing industry, and, much to karma’s delight, the oil refining industry as well. Needless to say, the town’s fortune dwindled and its people were left starving (fishing provided the main source of food) and furious. 
The sulfuric acid released into the water spread….
and spread…..
and spread.
The high acidity (sulfuric acid has a ph of 1) of the refinery waste caused ocean waters nearer to the coastline of Hambry to decrease significantly. Though such marine conditions did not alter fish edibility or survival, something happened to the waters of Clean Sea….

I is ugly guppy. 😦
Yeah, that happened… Freshwater guppies, the only fish species that seemed to be affected by the acidified water, began to mutate to morbidly obese proportions and acquire the face of squidward. The guppies then proceeded to flip the food chain around by eating whole jellyfish, tuna fish, swordfish, narwhals, and sharks.
However, due to its diverse marine and coastal wildlife, Hambry attracted scientists, environmentalists, and other professionals in the scientific line of study over the years. Among the brightest (aka least modest) were the deceitful marine biologist named Roland and self-righteous biochemist named Susan. Susan and Roland wanted to help the poor people of Hambry (aka retain their fame). Thus both worked together to develop a long-standing solution to rid the Clean Sea of its squidward guppies.

They decided that the most effective solution would be to neutralize the sulfuric acid and collect the resulting precipitate. In order to prescribe the correct amount of base solution to neutralize the waters near the coast, they needed to find the amount of sulfuric acid that was disposed of. Unfortunately, because of the poor planning and procedures occurring in the refining factories, such information was not consistently recorded.

Zzzzzzzz
Susan (the biochemist) proposed to calculate the ph level of the coastal ocean water to determine the increase in acidity caused by the sulfuric acid. She used the basic logarithmic equation ![]() , in which p(t) represents the actual ph level and the variable t represents the hydronium ion concentration in moles per liter. From there, the two measured the concentration of hydronium ion, and exponentialized and scientifically notated it to become:
, in which p(t) represents the actual ph level and the variable t represents the hydronium ion concentration in moles per liter. From there, the two measured the concentration of hydronium ion, and exponentialized and scientifically notated it to become:
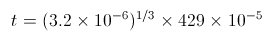 Roland, being the deceitful and treacherous jerkwad he was, had long wished to snatch the glory and adoration to be gotten from presenting such a solution to Hambry… So it would not be out of his character to tranquilize Susan and make off with her hard-earned work in a wild dash fueled by greed. And he did exactly that, except he took her Ti-84 and laptop as well.
Roland, being the deceitful and treacherous jerkwad he was, had long wished to snatch the glory and adoration to be gotten from presenting such a solution to Hambry… So it would not be out of his character to tranquilize Susan and make off with her hard-earned work in a wild dash fueled by greed. And he did exactly that, except he took her Ti-84 and laptop as well.
Susan, fueled by the passion to kick 300 Spartans into a bubbling pit of the underworld, had no intention of letting Roland seize victory over her work. Her hardy memory, improved by the sophomoric years of recalling lengthy chemistry formulas to the monster years of university spent cramming in chemical processes on a molecular level, allowed her to easily remember the value of t (hydronium ion concentration). However, she soon realized after the disappearance of her calculator and laptop that 1: Apple won’t insure the loss of the laptop and 2: she would not be able to easily calculate the logarithmic values.

Boohoo
The aboriginal village people conformed to an irrational and nonsensical religion in which they could act excessively condescending towards non-believers (esp. scientists). The more educated folks, including fellow scientists and the like, quickly fled from the village for fear of air contamination sickness. With no one left to turn to for the aid of modern technology, Susan sprinted desperately to the local library.
She made directly for the maths section of the library…
made a quick turn to the second row…
and dramatically lunged for the topmost grandpa-of-a-math-textbook. (Resulting in a twisted ankle and chipped glasses.)

Finally, after several seconds of flipping through pikachu doodles, she found the logarithm table. Her well-toned cerebrum from years of what seemed to be counter-intuitive arithmetic practice allowed her to WHAZAPP through the tedious decimal multiplication with ease.

Meep.
Susan first structured the equation by replacing the value t (representing the hydronium ion concentration) in the ph equation ![]() with her discovered t value, which was
with her discovered t value, which was ![]() .
.
Thus, her starting equation was:
She then demolished the power values by using the exponent log identity![]() and expanded the parenthesized values using the multiplication log identity
and expanded the parenthesized values using the multiplication log identity ![]() . She also broke down the value 429 into 4.29 x 100 so that the log value could be found on her logarithm table. The equation became even more grotesque:
. She also broke down the value 429 into 4.29 x 100 so that the log value could be found on her logarithm table. The equation became even more grotesque:
Now, she simply converted the multiplied log values into additions using the multiplication log identity once again, and flicked the negative six exponent by again using the exponent log identity. The equation became a massive obese blob:
At this point, Susan replaced the log 10 values to their whole number equivalents and began running her finger down the rows and columns of decimals to solve the log 3.2 and log 4.29 values. Her terrible short-sightedness caused her to skip repeatedly over the necessary log answers. In the end she whipped the crap out of the damn book with her handy dandy highlighter. From then on, the remaining steps were pretty self-explanatory. This is what she calculated (in her mind. Get yo nasty hands off of that TI 84):
Susan concluded that the Clean Sea near the coast of Hambry had a ph level of approximately 4.2. Oceanic fish start to die when the seawater has a ph of around 3 or less. Thus, Susan discovered that although fish other than the bloated guppies did not seem to experience side effects yet, they were at high risk of poisoning because of such a high level of water acidity. This would greatly compromise the health of Hambry villagers if one were desperate enough to eat one, and ultimately cast the people into famine. (Others would later check her work on a calculator and find out that her answer was correcto!)

After her breakthrough discovery, the bright lights of fame shined upon Susan, which she later used to amass a fortune. Before, she was loathed by the villagers due to a poisonous combination of jealousy, sexism, religion-induced xenophobia, and archaic prosophobia. However, after her success, most villagers came to realize and respect the importance of technology and science and how awesome females are. Thus, they threw out their thousand-year-old religion and quickly adopted a new ideology which greatly empowered women both economically and politically. After Susan cooperated with other scientists to diagnose and create an appropriate base solution to eradicate ocean acidity severity, Hambry experienced a dramatic swell in economic growth. This was largely due to the fact that Hambry’s new ideology created room for greater industrial and commercial prospects. In fact, the town expanded and leached onto the entire left side of the Clean Sea that it became informally known as ‘Clean Hambry Thanks to Susan’ City. Most importantly, the semi-city implemented much stricter safety regulations for factory conduct, adopted greener renewable energy sources, and ultimately reduced air and water pollution levels significantly.

Had Roland calculated the logarithmic values of the plagiarized equation, he would certainly have beat Susan to the prize. (Un)fortunately, he accidentally divided by zero, causing a black hole to poof into existance in his heart, sucking him into an endless oblivion. 😦

FIN.
Logarithms were very useful in making calculations such as exponentials less laborious when there were few/no calculators. This is because using the laws of logarithms until the problem consists of whole numbers and logs, each corresponding logarithm value could be found on a log table, thus making the process of solving calculations involving logs much easier.
Usually, results are accurate to the fourth or fifth decimal place, due to the fact that most log tables available are limited to this many decimals representing accuracy. However, this can still be considered reasonably accurate, as the convenience in log tables had advanced numerous fields of science including astronomy. Thus, accuracy of such log values in a logarithm table with at least four decimal places seemed not to deter progress in science this way.
In the past before such logarithm tables were created, multiplication was used in prosthaphaeresis and other mathematical methods involving trig identities. For example, ships in the sixteenth century of Europe, especially those on lengthy voyages, required mathematical positioning of astronomical objects in the sky to find and keep constant track of their course. Thus, astronomers would apply the trigonometric equations of prosthaphaeresis to spherical trigonometry, which were used to compute models of star and planet positions. However, the creation of logarithms (and log tables) made the task less tedious.


















